- Home
- About Us
- Work
- Journal
- Contact
- Uninstall need for speed shift
- Tekken 6 free download for pc full version
- The witness bird calls
- The lost vikings controls
- Install android studio ubuntu
- Star wars knights of the old republic builds
- Coolmuster iphone backup extractor serial
- Structure of an atom
- Propresenter 5
- 136-1 hz tuning fork benefits
- Home
- About Us
- Work
- Journal
- Contact
- Uninstall need for speed shift
- Tekken 6 free download for pc full version
- The witness bird calls
- The lost vikings controls
- Install android studio ubuntu
- Star wars knights of the old republic builds
- Coolmuster iphone backup extractor serial
- Structure of an atom
- Propresenter 5
- 136-1 hz tuning fork benefits

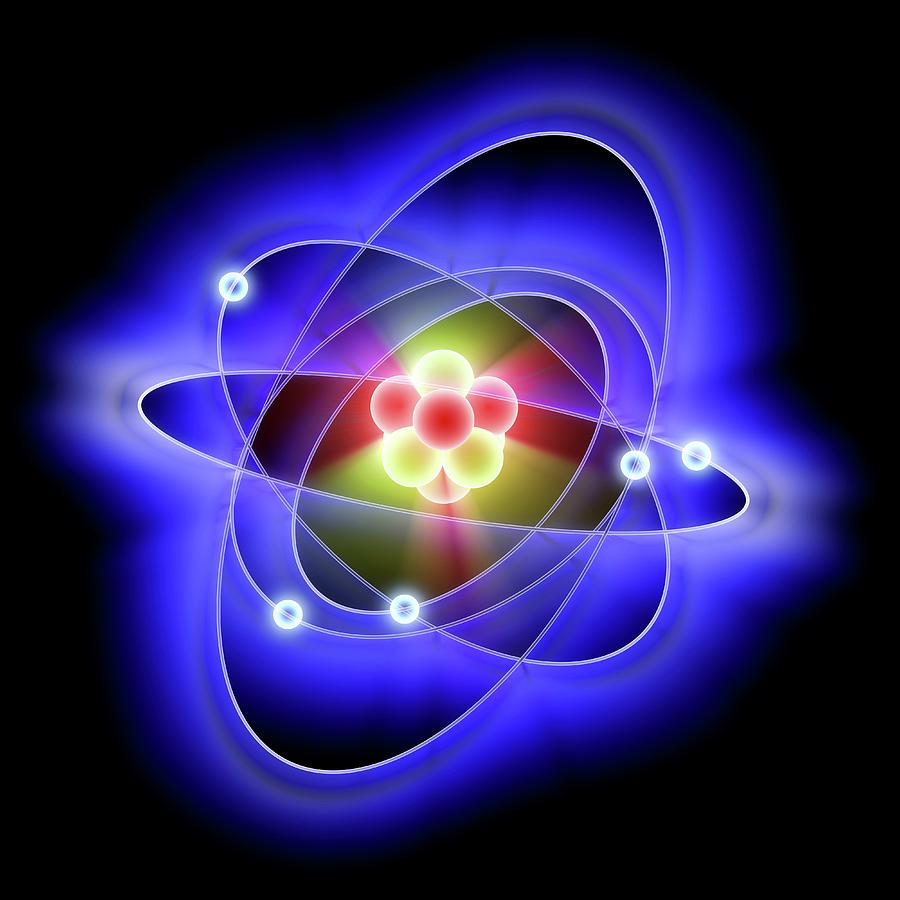
The alpha particles which deflected through large angles were those which passed close to the positive body. The alpha particles which deflected through small angles were those which passed at a distance pretty far from this positive body. As some of the alpha particles were deflected by small angles, and since alpha particles are positively charged they could be deflected only by some other positively charged body present within the atom.Since most of the alpha particles passed through the foil without undergoing any deflection, there must be sufficient empty space within the atom.through an angle greater than 90 degrees.įrom these observations, Rutherford drew the following conclusions:. only one in 12000 were deflected back i.e. Some particles were deflected through small angles and a few were deflected through large angles.Most of the particles that are 99.9% of the particles passed through the foil without undergoing any deflection.Wherever alpha particles struck the screen a flash of light is produced at that point on the screen.įrom these experiments, Rutherford made the following observations:. He observed the scattering of the rays after hitting the foil by placing a circular zinc sulfide screen around the metal foil. These were obtained from radium placed in the cavity of block of lead and made into a fine beam with a slit. Rutherford’s scattering experiment: In 1911, Rutherford performed an experiment in which he bombarded a thin foil of a heavy metal like gold with a beam of fast-moving alpha particles which are doubly ionized helium ions or He2+ ions, each having 2 units of positive charge and 4 units of mass i.e. So, the atom as a whole is electrically neutral.Īlthough Thomson’s model explained that the atom is electrically neutral, the result of experiments carried out by other scientists could not be explained by this model. The negative and positive charges are equal in magnitude.An atom consists of a positively charged sphere and the electrons are embedded in it.This model can be compared with a watermelon in which the seeds are embedded. It is like a Christmas Pudding with dry fruit. Thomson proposed that an atom was a sphere of positive charge in which a number of electrons were embedded, which were sufficient to neutralize the positive charge. In the year 1904, J.J Thomson was the first one to propose a model for the structure of an atom. How these subatomic particles are arranged in an atom? In this blog, we will learn about this arrangement of subatomic particles in an atom i.e.

It consists of two fundamental particles, namely, electrons and protons. By the end of the 19th century, it was discovered that the atom is not an indivisible particle.
